|
Services: Medical Devices & Supplies
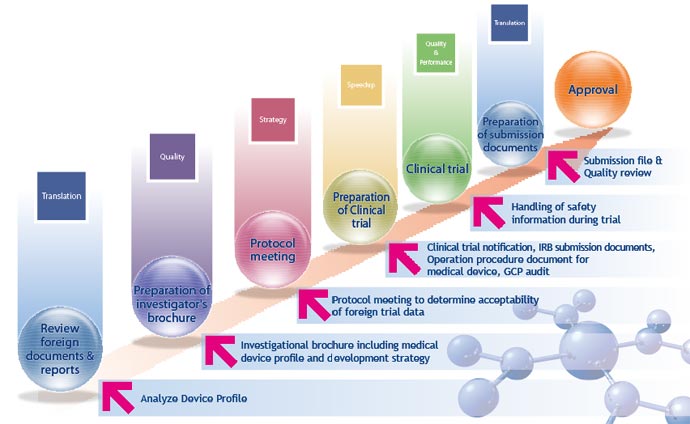
Pharma Training consultants work with clients to obtain approval for the development of medical devices and supplies in Japan. We
assist clients with all phases of the approval process:
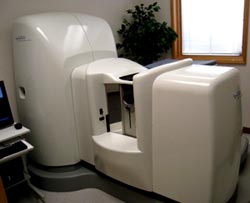
Pharma Training has consulted with clients to bring innovative products to Japan, including a new automated breast imaging system. Click here to view of demonstration of this device.
- If the medical device was approved by the FDA and/or EMEA (or other authority)
prior to MHLW, or is currently undergoing trials outside of Japan, we
will read and review an investigational plan, preclinical study reports,
marketing analysis documents, and regulatory documents so that we can
understand the device profile (description, clinical
performance, safety, sterilization, stability & durability, and risk
analysis) within a confidential agreement with the client.
- Pharma Training will create an investigator's brochure in Japanese as a
scientific document of the medical device (an investigational plan may be
available if needed). This document, outlining the medical device profile and development strategy,
is of critical importance to the Japanese Authority. We will therefore conduct a quality check on the consistency between the
study data and investigator's brochure (or investigational plan) for the
submission documents to the Japanese Authority and Investigators.
- A client who intends to get the approval of their medical device from the
Japanese Authority will have a PMDA meeting regarding the acceptability of the
preclinical and clinical trial data obtained outside of Japan. If
a client has to add a clinical trial with Japanese patients along with the
international clinical study results, Pharma Training will offer the
regulatory strategic plan including Japanese trial protocol as well as the Regulatory Strategy Solution. For a standard medical device, a Japanese trial may
not be needed for a submission to the Japanese Authority; for an
innovative device or combination device with drug, clients are required to perform a
Japanese trial with a small sample size.
- Pharma Training will support the preparation of clinical trials in Japan, including clinical trial notification (CTN),
Institute IRB submission (with Q&A), operational procedure documents, and GCP audit with the audit
timing to be conducted according to SOP and GCP.
- Pharma Training will support clients in handling serious adverse
event information of the medical device during trials in Japan in compliance
with Pharmaceutical Affairs Law as well as ICH-GCP of the pharmaceutical
product.
- Pharma Training will support clients in preparing the submission
regulatory documents. Following submission, Pharma Training will support the quality review (or
inspection) by the Japanese Authority so that a client can ensure the quality assurance covering clinical data
(under GCP) and preclinical data (under GLP).
|
|