|
||
 |
||
| Overview | Regulatory Strategic Solution | Biopharmaceutical Product CMC | Clinical Trial Service | PET Drugs | Pharmaceutical Product Solutions | Medical Devices & Supplies | ||
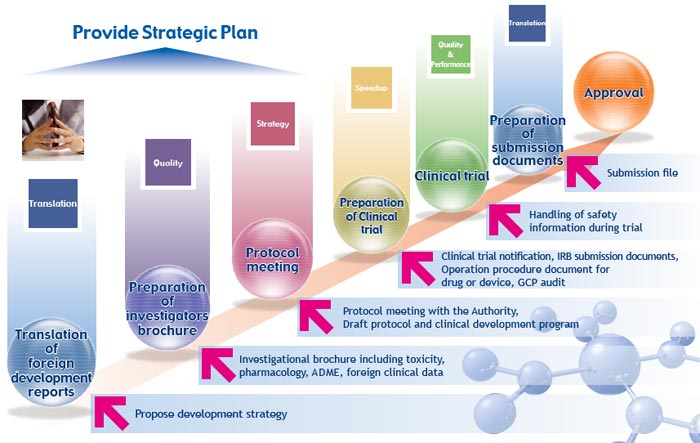
| Services: Regulatory Strategic Solution
Pharma Training consultants will create and offer a regulatory strategic plan and promote clinical trials for approval of your products in Japan. First, we create a Regulatory Affairs Strategy which addresses key issues such as:
 |
||